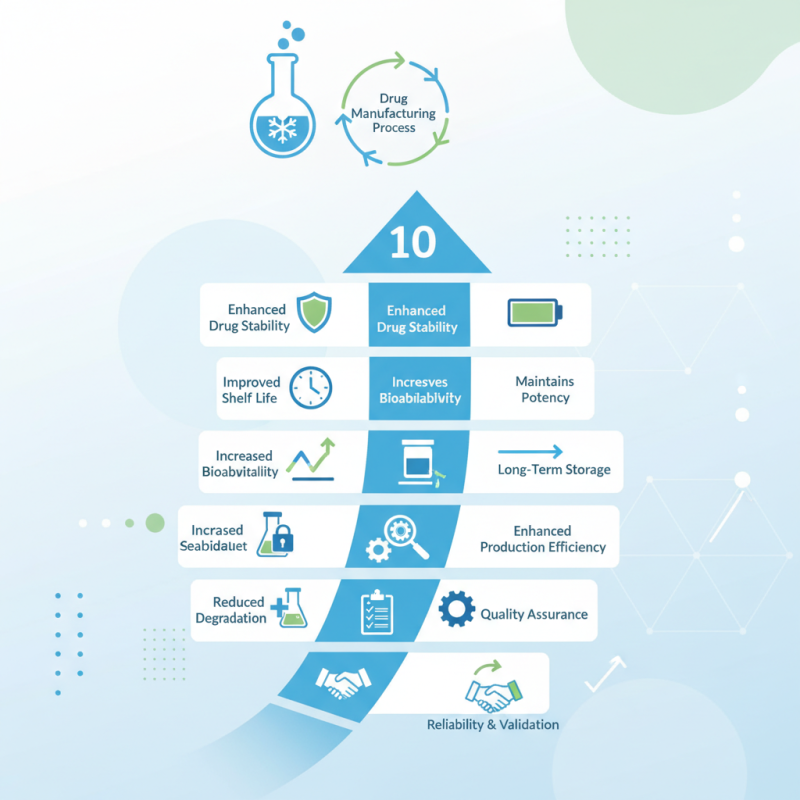
Top 10 Benefits of Using a Pharmaceutical Lyophilizer in Drug Manufacturing?
In the realm of drug manufacturing, the importance of a Pharmaceutical Lyophilizer cannot be overstated. These advanced machines play a crucial role in the freeze-drying process, significantly enhancing the stability of pharmaceuticals. They help preserve sensitive compounds, making them more effective for patients.
The benefits of using a Pharmaceutical Lyophilizer extend beyond mere product preservation. They contribute to improved shelf life and increased bioavailability. This technology minimizes degradation, allowing for longer-term storage without losing potency.
However, not all lyophilizers are created equal. Selecting the right Pharmaceutical Lyophilizer involves understanding specific manufacturing needs and product requirements. Quality assurance and validation are essential to ensure reliability. Each decision impacts production efficiency and overall success in the pharmaceutical industry.
Introduction to Pharmaceutical Lyophilizers and Their Importance
Pharmaceutical lyophilizers play a vital role in the drug manufacturing process. These devices freeze and dry substances, ensuring that the active ingredients in pharmaceuticals remain stable over time. By removing moisture, lyophilization helps preserve the potency and efficacy of drugs. This process is especially crucial for biologics, which can be sensitive to environmental changes.
The importance of lyophilizers extends beyond stabilization. They contribute to the overall quality of the drug product. A well-executed lyophilization cycle can prevent degradation, which is critical for maintaining patient safety. However, not all pharmaceutical companies have mastered this technique. Some face challenges with process consistency, leading to variable product quality. Continuous improvement is necessary for these companies to achieve reliable results in their manufacturing processes.
Moreover, the choice of parameters during lyophilization can significantly affect the final product. Temperature, pressure, and time must be optimized to ensure efficient drying. Mistakes made in this phase can result in poor quality products that do not meet regulatory standards. Industry experts stress the need for comprehensive training and ongoing research to address these challenges. By focusing on these aspects, manufacturers can ensure the reliability and efficacy of their pharmaceutical products.
Enhanced Stability and Shelf Life of Pharmaceuticals through Lyophilization
Lyophilization, or freeze-drying, significantly enhances the stability of pharmaceuticals. By removing moisture, this process protects active ingredients from degradation. Reduced water content also minimizes the risk of microbial growth. Consequently, drugs can maintain their potency and efficacy over extended periods, making lyophilizers invaluable in drug manufacturing.
The shelf life of lyophilized products is often extended beyond that of traditional formulations. Many pharmaceuticals that are sensitive to heat or humidity benefit greatly from this process. Manufacturers can store these products at room temperature, easing transportation and storage challenges. Furthermore, the reconstitution of lyophilized drugs typically results in a solution that is just as effective as the original.
However, the process is not without its challenges. Achieving optimal conditions for freeze-drying requires extensive knowledge and experience. Inconsistent results can arise if parameters are not closely monitored. This aspect highlights the importance of skilled personnel in ensuring the quality and reliability of lyophilized products. Proper training and adherence to guidelines are essential for success in this sensitive area of pharmaceutical manufacturing.
Improved Product Quality and Bioavailability in Drug Manufacturing
Lyophilization, or freeze-drying, plays a crucial role in drug manufacturing. It enhances product quality significantly. According to a report by the American Pharmaceutical Review, products processed through lyophilization show improved solubility. This leads to better bioavailability in the body. Bioavailability is vital for patient outcomes. If a drug isn’t absorbed well, its efficacy is diminished.
Furthermore, lyophilizers help stabilize complex formulations. They prevent degradation, ensuring active ingredients remain intact. A study published in the Journal of Pharmaceutical Sciences noted that lyophilized products have a shelf-life increase of up to 24 months. This is especially important in emergencies, where access to medications can be critical. However, lyophilization can be complex. Process optimization requires expertise and experience.
Challenging conditions during the freeze-drying process can lead to variability. This variability may affect the final product's performance and patient safety. Continuous monitoring is essential. By improving product quality and bioavailability, lyophilizers play a vital role in the future of pharmaceuticals. As the demand for efficient drugs rises, so does the importance of this technology.
Cost-Effectiveness and Efficiency of Lyophilized Drug Production

In pharmaceutical drug manufacturing, cost-effectiveness is pivotal. Using lyophilization can significantly reduce production expenses. This method removes water from drugs, extending their shelf life. As a result, pharmaceuticals become more stable and easier to transport, lowering storage costs.
Efficiency is another critical factor. Lyophilized products can be produced in large batches. This scalability allows manufacturers to meet market demands quickly. The process also minimizes waste, enhancing resource use. However, it's essential to monitor the lyophilization process closely to avoid compromising drug quality.
Tips for optimizing lyophilization include ensuring proper temperature control. Even minor fluctuations can affect outcomes. Regular maintenance of equipment can prevent unexpected downtimes. Take time to train staff thoroughly on operational protocols. Ensuring everyone understands the process is crucial for success.
Versatility and Adaptability of Lyophilizers in Various Drug Formats
Lyophilizers, or freeze dryers, offer remarkable versatility in drug manufacturing. These machines adapt to various drug formats, including vaccines, biologics, and small molecules. Industry reports suggest that the global lyophilization market is projected to reach around $4.7 billion by 2027, driven by increasing demand for sterile products.
Lyophilization enhances stability and extends shelf life. By removing moisture, it prevents degradation. This process allows for better preservation of sensitive compounds. Specialized lyophilizers can handle different vial sizes and formulations, ensuring flexibility. However, not all products benefit equally from this method. Some drugs may experience changes in efficacy.
Tips: Always conduct small-scale trials to understand how lyophilization affects your specific product. Pay careful attention to formulation stability during the freeze-drying process.
The adaptability of lyophilizers can lead to challenges. Variability in the physical state of the drug may impact performance. Moreover, optimizing cycle parameters requires in-depth knowledge. This can create hurdles for manufacturers new to lyophilization. Investing in staff training can mitigate these issues and ensure effective operations.
Top 10 Benefits of Using a Pharmaceutical Lyophilizer in Drug Manufacturing
| Benefit | Description | Drug Formats |
|---|---|---|
| Increased Stability | Lyophilization significantly improves the stability of sensitive pharmaceuticals. | Injectables, Biologics |
| Improved Solubility | Lyophilized products can offer enhanced solubility upon reconstitution. | Powders for Injection, Oral Formulations |
| Extended Shelf Life | Lyophilization can extend the shelf life of drugs by reducing moisture content. | Injectables, Vaccines |
| Reduced Transportation Costs | Lightweight lyophilized products lower shipping expenses. | Powdered Drugs |
| Versatile Applications | Applicable to a wide range of drug types including proteins and peptides. | Proteins, Peptides |
| Scalability | Ease of scaling up processes from lab to commercial production. | All Drug Formats |
| Enhanced Product Purity | Fewer impurities through controlled drying processes. | Pharmaceuticals, Biologics |
| Compatibility with Various Formulations | Can be integrated with various excipients and formulations. | All Forms, Including Complex Formulations |
| Promotes Targeted Delivery | Improves drug delivery systems by preserving bioactivity. | Targeted Therapies |
| Customizable Processing | Parameters can be tailored to fit specific product needs. | Various Drug Types |
Related Posts
-

Top 10 Pharmaceutical Lyophilizer Models You Should Know?
-

2026 Best Biopharma Production Techniques and Innovations to Watch?
-

Ultimate Tips for Efficient Peptide Synthesis Machine Use?
-

10 Essential Tips for Optimizing Biopharma Manufacturing Efficiency
-
Top Tips for High Performance Liquid Chromatography Techniques and Applications
-

Top 10 Trends Shaping Biopharma Manufacturing Today?
